 Figure
1 . Copper mine.
Figure
1 . Copper mine.Thiobacillus:
A Microbial Miner
Environmental
Microbiology
Experiments for middle and high school
List
of experiments
 Figure
1 . Copper mine. Figure
1 . Copper mine. |
Thiobacillus:
A Microbial Miner |
|
| Objectives |
|
||
|
Background |
For thousands of years, refiners have been extracting copper from low-grade copper ore by a process called leaching (Figure 1). Until 1957, however, nobody realized that the extraction would be impossible without the bacterium Thiobacillus ferrooxidans, which thrives in acidic environments such as mining sites. Thanks to this microbe, millions of tons of copper are recovered from billions of tons of low-grade ore every year. Thiobacillus ferrooxidans is one of several species of chemoautotrophic bacteria, which are becoming increasingly important in the extraction of a variety of metals. The chemoautotrophs obtain their energy from the oxidation of inorganic substances (iron and sulfur, in the case of T. ferrooxidans). This allows them to stimulate reactions involving minerals that are unaffected by, and may even be poisonous to, other bacteria. Moreover, chemoautotrophic bacteria thrive in the absence of organic matter because they extract their carbon dioxide directly from the carbon dioxide in the atmosphere. The bacteria are naturally present in the leaching solution prepared at the mining site. The refiners grind the copper-bearing rock and heap it into a pit, called a dump. Then they pour water mixed with sulfuric acid (the leach solution) through the dump. When the leach solution runs out through the bottom of the dump, it contains copper sulfate (CuSO4) in solution. The refiners then add metallic iron to the leach solution. The iron reacts with the copper sulfate to release metallic copper. (See figure 2). Thiobacillus frees copper from the ore in a variety of ways. Most commonly, the bacillus reacts with iron and sulfur compounds that are in the rock. The rock is likely to contain compounds of iron and sulfur, such as iron pyrite (FeS2). As the leach solution trickles down through the ore-bearing rock, T. ferrooxidans oxidizes the Fe2+ ion by removing an electron from it. This turns the ferrous ion into a ferric ion, Fe(III). The sulfur that was in the FeS molecule can now combine with hydrogen ions and oxygen molecules to form sulfuric acid (H2SO4). In addition to the iron pyrite, the ore contains compounds of copper and sulfur, such as CuS. The ferric ion oxidizes the monovalent copper ion, Cu(I) to create a divalent copper ion, Cu(II), which then combines with the sulfate ion, contributed by the sulfuric acid, to form copper sulfate (CuSO4). Now the leach solution has copper sulfate dissolved in it, the refiners add metallic iron. The iron reacts with the copper sulfate to form ferrous sulfate and metallic copper. Thiobacillus can now reoxidize the ferrous ion to ferric ion. The mining industry has begin to use chemoautotrophic bacteria to retrieve other elements that are present in low concentrations in ores. Thiobacillus is being used to extract cobalt at the Idaho National Laboratory, and U.S. Gold Corporation uses Thiobacillus to free gold from rock. Microbial mining offers a possible solution to another problem that threatens modern industrial societies-pollution of the soil and groundwater by unextracted metallic ions in waste water. Chemoautotrophic bacteria that can take up and accumulate toxic metal ions in their cells may be useful in removing metal pollutants in waste water. |
||
| Materials | Beakers (22) Copper turnings 0.5M ferric oxide CaCO3 (calcium carbonate) Tubes containing Thiobacillus broth, 6 mL per tube (2) Thiobacillus thioparus (Carolina Biological Supply) pH paper |
||
| Procedure Part A: Oxidation of copper |
1. Label two beakers: one "ferric ion" and the other "distilled water." 2. Weight two equal samples of copper turnings. Transfer the samples to the labeled beakers. 3. Add 0.5M ferric oxide solution to the beaker labeled "ferric ion." Add an equal volume of distilled water to the beaker labeled "distilled water." 4. Observe the two beakers each day for one week. Record your observations. Note the color of the solution, the condition of the soil, and the quantity of the solid. |
||
| Procedure Part B: Growing Thiobacillus thioparus |
1. Add 0.1 g powdered CaCO3 to one tube of Thiobacillus broth. Label the tube "buffered." 2. Obtain a second tube of Thiobacillus broth and label it "not buffered." 3. Using pH paper, check the pH of the Thiobacillus broth in each tube. Record the pH as the starting pH. 4. Inoculate each tube prepared in steps 1 and 2 with 2 mL of Thiobacillus culture. Incubate the tubes at 30°C. 5. Observe the two tubes daily for two week. Record the pH and condition of the broth. |
||
| Questions |
|
||
| Source | Applications in Biology/Chemistry: Microorgaisms CORD Communications. | ||
Figure
1. Biological leaching of copper ores.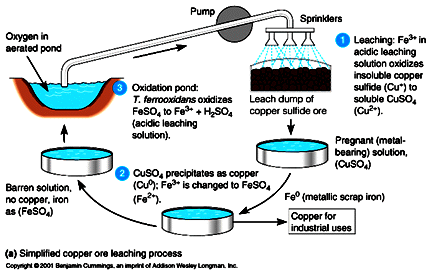 |